AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes. The atomic number of an element is the number of protons in the nucleus of an atom of that element. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. The periodic table is a tabular array of the chemical elements organized by atomic number, from the element with the lowest atomic number, hydrogen, to the element with the highest atomic number, oganesson. For instance they discovered phosphorus when they isolated it from urine. Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns (called groups) according to increasing atomic number. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. Several other attempts were made to group elements together over the coming decades. Each next element in a period has one more proton and is less metallic than its predecessor. 
All elements in a row have the same number of electron shells. Assess students understanding of prior learning by revisiting the topic of atomic structure. Quiz your students on periodic table trends, such as melting points, ionisation energies and atomic radiuses. The term is also spelled ionisation energy (British English). A period on the periodic table is a row of chemical elements. In the context of chemistry and the periodic table, periodicity refers to trends or recurring variations in element properties with increasing atomic number. Help post-16 students consolidate their ideas about atoms and atomic structure by working in pairs to identify key concepts. 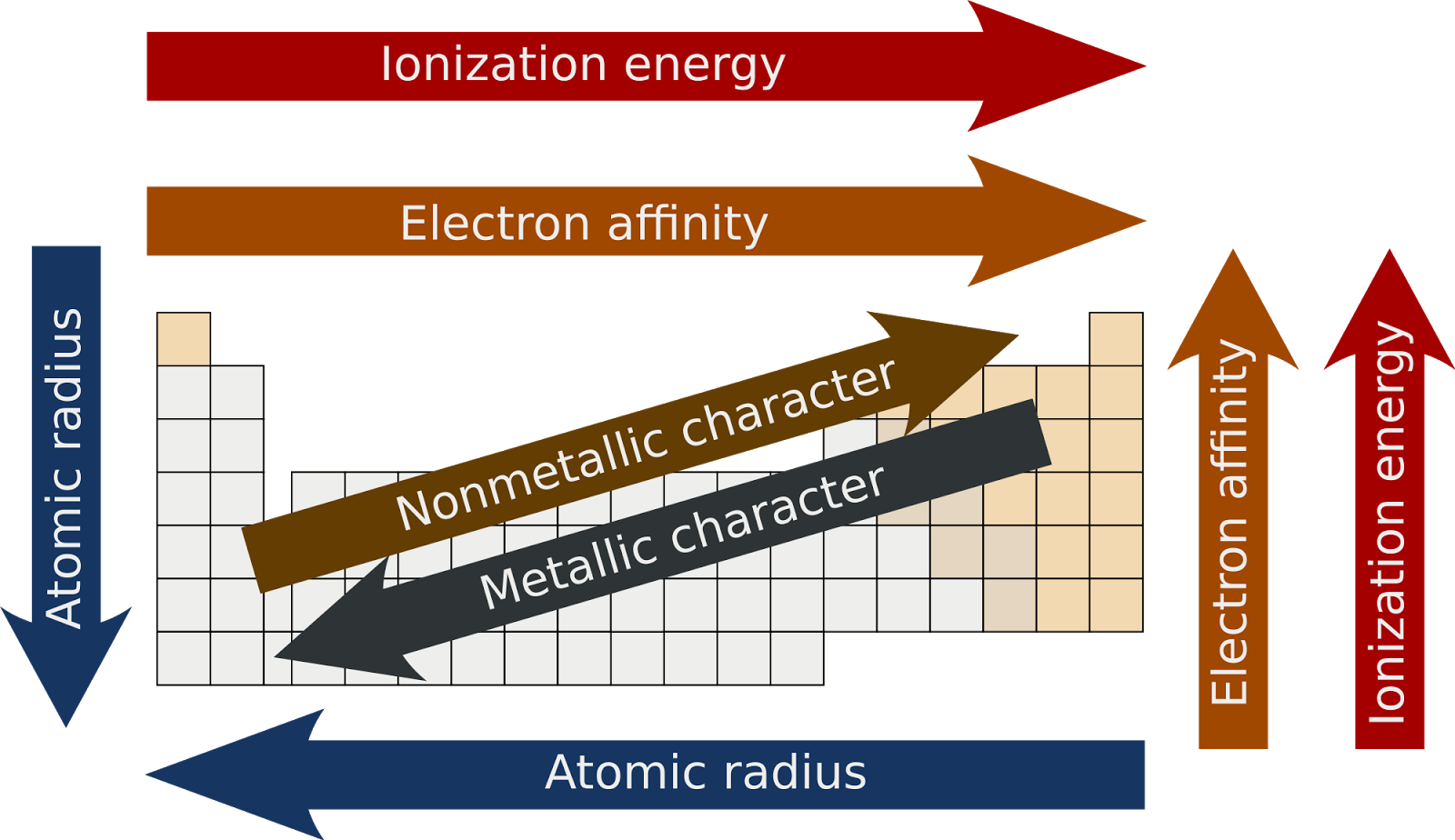
By definition, ionization energy is the minimum energy needed to remove the most loosely bound electron from a gaseous atom or ion. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. It is a periodic table trend that increases moving across the table and decreases moving down it. /periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
